You're at the pharmacy counter and the pharmacist tells you they have a generic version of your prescription. It's significantly cheaper, but a small seed of doubt plants itself: Is it actually the same thing? Will it work as well? You might have heard a friend say they felt "different" after switching, or perhaps you've read a scary headline about "pill quality." The truth is that while the vast majority of people switch without a single issue, the science isn't a simple "yes" or "no" for every single drug.
The Gold Standard: How Bioequivalence Works
To understand if a generic is "the same," we have to look at how the FDA (U.S. Food and Drug Administration) regulates them. They don't just take the manufacturer's word for it; they require proof of bioequivalence. In plain English, this means the generic drug must deliver the same amount of active ingredient into your bloodstream within the same timeframe as the brand-name version.
The technical bar is high: the drug's performance must fall within a 90% confidence interval between 80% and 125% of the brand-name product's pharmacokinetic parameters. While a 20% variability might sound like a lot, for most medications-like the ones you take for high cholesterol or common infections-your body doesn't notice the difference. These are drugs with a wide therapeutic window, meaning the dose can fluctuate slightly without changing the outcome.
Where the Risk Increases: Narrow Therapeutic Index Drugs
Here is where we need to be specific. Not all drugs are created equal. Some fall into a category called Narrow Therapeutic Index (NTI) drugs. For these medications, the line between a dose that works and a dose that is toxic (or ineffective) is incredibly thin. Small variations in blood concentration can lead to therapeutic failure.
A prime example is Phenytoin, used to treat seizures. Research has shown that generic versions of phenytoin can have plasma drug levels 22% to 31% lower than the brand-name version. In a world where a 10% drop could mean the difference between a seizure-free day and a trip to the ER, that gap is a big deal. This is why the American Academy of Neurology suggests that patients with unstable epilepsy may need closer monitoring or should stick to the brand name.
| Drug Class | General Research Consensus | Key Risk/Benefit |
|---|---|---|
| Statins (e.g., Atorvastatin) | Generally equivalent or better | Lower hazard ratios for death in some large studies |
| Antiepileptics (e.g., Levetiracetam) | Higher variability | Risk of breakthrough seizures and mood swings |
| Anti-hypertensives (e.g., Amlodipine) | Mostly equivalent | Some reports of increased side effects; others show better adherence |
| NTI Drugs | Caution required | Small changes in blood levels can cause toxicity or failure |

The Evidence: Comparing the Big Studies
If you look at the data, the results are often contradictory because they look at different things. For instance, a 2017 study in *Circulation* found that patients switching to generic blood pressure meds like amlodipine saw a 12.3% increase in adverse events. It sounds worrying, right? But then look at a massive 2020 study from *Nature Scientific Reports* that tracked 8.5 million people in Austria. After adjusting for variables, they found that generic versions of 10 out of 17 common drugs (including those for diabetes and heart failure) were actually associated with fewer deaths.
Why the difference? It often comes down to the "switching effect." A 2022 study in *JAMA Neurology* followed over 112,000 epilepsy patients and found that while the risk of emergency room visits jumped by 12.7% immediately after switching to a generic, that risk disappeared after 90 days. Your body often needs time to adapt to a slightly different formulation, even if the active ingredient is the same.
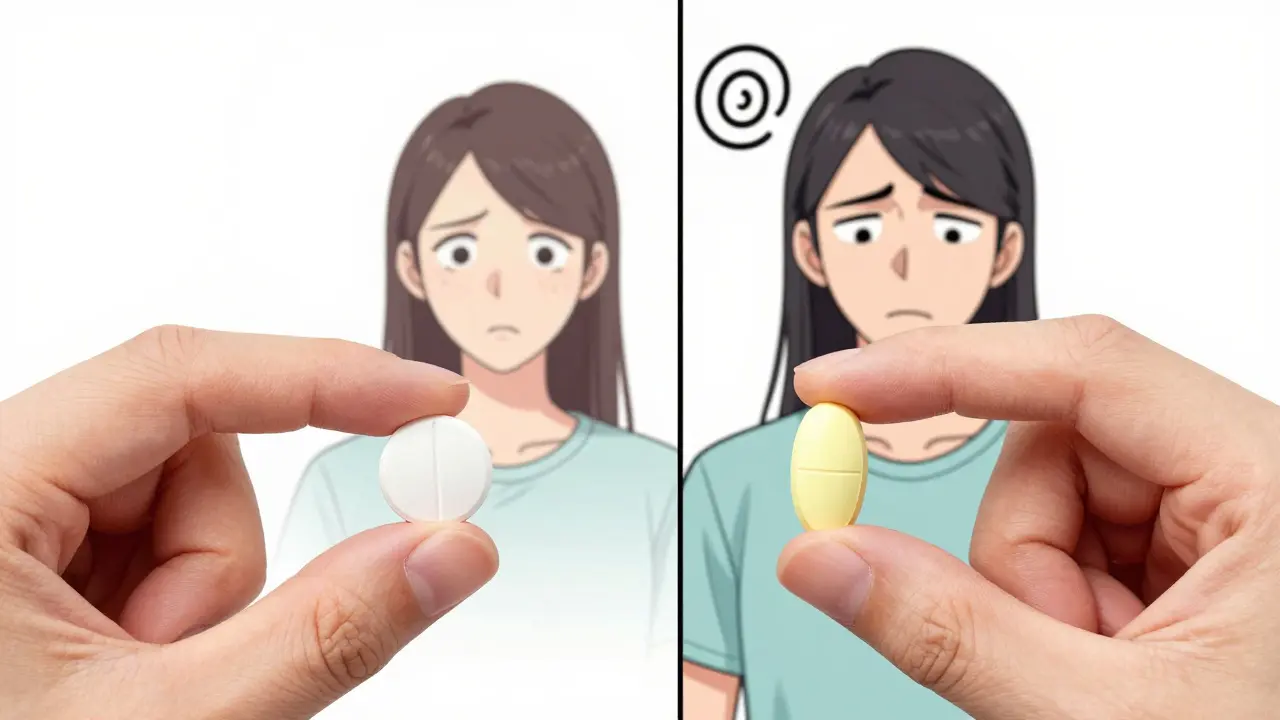
The "Generic-to-Generic" Trap
One of the most overlooked problems isn't switching from brand to generic-it's switching from one generic manufacturer to another. Many pharmacies change their generic suppliers to save costs. You might start the month with a small round white pill and end the month with an oval pale-yellow pill, even though the drug name is the same.
This causes a huge psychological and practical problem. A 2023 study found that 67% of patients identify their meds by the physical "trademark" (color, shape). When the pill looks different, patients get confused. In one study, only 19.7% of patients switching between generics actually knew why they were taking the medication. This confusion leads to "medication duplication," where a patient thinks they are taking a new drug when they are actually taking a second dose of the same thing.
How to Switch Safely: A Practical Guide
You don't need to be afraid of generics, but you should be informed. If you are taking a medication for a life-threatening condition or one with a narrow therapeutic window, don't just let the pharmacist make the call. You need a strategy.
- Ask about the "Window": Ask your doctor if your medication is a Narrow Therapeutic Index (NTI) drug. If it is, request therapeutic drug monitoring (blood tests) during the first few months of the switch.
- Log Your Symptoms: Keep a simple diary for 30 days after the switch. Note any new headaches, mood swings, or changes in efficacy. This helps your doctor distinguish between a side effect and a failure of the drug.
- Request a Consistent Manufacturer: If you find a generic that works perfectly, ask your pharmacy if they can stick with that specific manufacturer to avoid the "generic-to-generic" shuffle.
- Verify the Indication: Don't rely on the pill's color. Read the label every time to ensure you know exactly what the drug is for and why you're taking it.
The cost of a mistake can be higher than the savings. For example, managing a breakthrough seizure caused by a bad generic switch can cost nearly $2,000 per incident. If you're in a high-risk category, the brand-name price tag is a small price to pay for stability.
Are generic drugs exactly the same as brand-name drugs?
They are chemically equivalent in terms of the active ingredient, but they can differ in "inactive ingredients" like fillers, binders, and dyes. While the FDA ensures they are bioequivalent (meaning they work the same way in the blood), these inactive ingredients can occasionally cause different side effects or affect how some people absorb the drug.
Why do some people feel worse after switching to a generic?
This can happen for two reasons: the "nocebo effect," where the expectation that a cheaper drug is inferior creates real physical symptoms, or actual sensitivity to the inactive ingredients. In rare cases, specifically with NTI drugs, the slight variation in absorption allowed by FDA standards (80-125%) can be enough to push a patient's blood levels outside the effective range.
Which medications should I be most cautious about when switching?
Be most cautious with antiepileptics (like phenytoin or levetiracetam), certain cardiovascular medications, and any drug labeled as having a Narrow Therapeutic Index. If your condition is unstable or requires precise dosing, consult your specialist before switching.
Can I ask my pharmacist to stop switching my generic brands?
Yes. You can request that the pharmacy uses the same manufacturer for every refill. While they may not always be able to guarantee it due to supply chain issues, pharmacies can often flag your account to keep the medication consistent.
How do I know if a generic is failing me?
Look for "breakthrough symptoms." If you were stable on a brand-name drug (e.g., no seizures, stable blood pressure) and you suddenly experience a return of symptoms shortly after switching, it may be a sign of bioinequivalence for your specific body. Contact your doctor for a serum drug level test.

