For millions of people, picking up a prescription feels like decoding a foreign language. You walk out of the pharmacy with a small bottle labeled esomeprazole, but you remember your doctor saying "Nexium." Was this a mistake? Is it weaker? Safer? Cheaper? If you’ve ever felt confused, you’re not alone. About 43% of Americans believe generic drugs aren’t as good as brand-name ones-even though the FDA says they’re required to work the same way. The problem isn’t the medicine. It’s the explanation.
What exactly is a generic drug?
A generic drug is not a copy. It’s not a knockoff. It’s the exact same medicine, just without the brand name. The FDA requires that a generic version has the same active ingredient, the same strength, the same form (tablet, capsule, liquid), and the same way it’s taken (by mouth, injection, etc.) as the original brand. If your brand-name pill is 20mg of esomeprazole, your generic must be 20mg of esomeprazole. No more, no less.
Think of it like buying cereal. You might reach for Cheerios because you know the taste. But store-brand oat cereal? It’s made the same way, with the same ingredients, and tastes nearly identical. The only differences? The box is simpler, and it costs half as much. That’s what generics do for medicine.
The key word here is bioequivalence. This means the generic must deliver between 80% and 125% of the active ingredient into your bloodstream compared to the brand. That’s not a wide gap-it’s a tight, science-backed range. The FDA tested this with over 11,000 generic drugs between 2010 and 2020. Only 1.3% failed. That’s not a fluke. That’s a standard.
Why do people still doubt generics?
Because they’ve been told lies-or at least, oversimplified truths.
Some guides say: "Generics are exactly the same." That’s misleading. They’re the same in what matters: the active ingredient and how your body uses it. But they’re not identical in every way. The color? Different. The shape? Different. The fillers? Sometimes different. Lactose instead of sugar. Cornstarch instead of talc. These don’t change how the drug works. But if you’re told "it’s exactly the same," and then you notice your pill looks different, you start to wonder.
There’s also the issue of "authorized generics." These are brand-name drugs made by the same company, but sold under a generic label. They’re 100% identical to the brand-same factory, same ingredients, same packaging. But they cost less. Most patients don’t know this exists. And when they switch from a brand to a regular generic and feel "different," they blame the generic-not the fact they switched from an authorized version.
And then there are the exceptions. Drugs like levothyroxine (for thyroid), warfarin (a blood thinner), and phenytoin (for seizures) have what’s called a "narrow therapeutic index." That means even tiny changes in blood levels can cause problems. For these, some doctors still prefer the brand. Not because generics don’t work-but because consistency matters. Switching between different generic makers? That’s where trouble can creep in. The FDA doesn’t ban these generics. But they don’t say "they’re all the same" either.
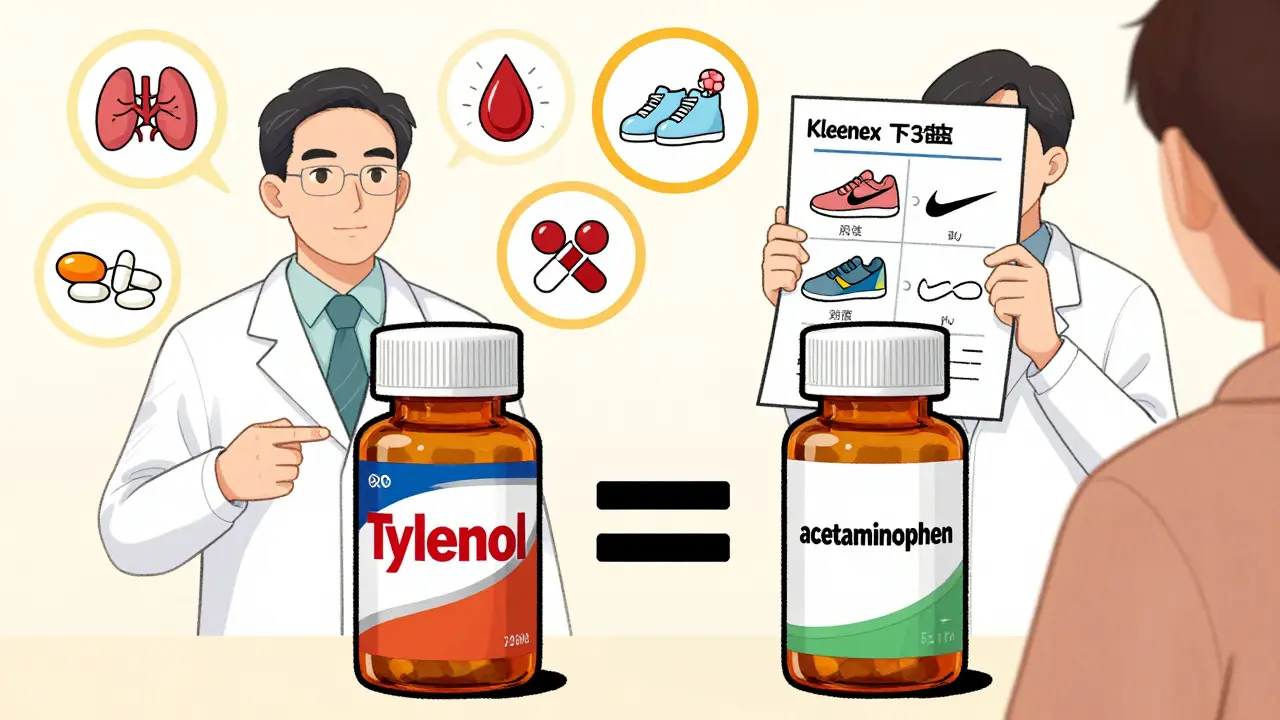
How do consumer language guides fix this?
They stop talking like a textbook and start talking like a human.
Old patient handouts? Written at a 10th-grade reading level. The FDA’s plain-language guides? Written at a 6th-grade level. That’s not dumbing down. That’s clarity. A 2021 study found that when patients got these simplified guides, their understanding jumped by 37 percentage points. That’s not a small win. That’s life-changing.
The best guides use analogies people already get:
- "Tylenol is to acetaminophen like Kleenex is to tissues."
- "Lipitor is to atorvastatin like Nike is to generic running shoes. Same function. Different brand. Same results."
They use side-by-side images: one bottle with "Nexium," the other with "esomeprazole." Same size. Same dosage. Same instructions. The only difference? The price tag.
They also include "teach-back" questions:
- "Can you tell me why your doctor switched you to this pill?"
- "What’s the active ingredient in your medicine?"
- "How much money will you save each month?"
Pharmacists trained with these guides spend just 90 seconds explaining. Three points: same active ingredient, same effect, same safety. And they save you an average of $387 per prescription every year.
What’s changing right now?
The FDA launched its "Generic Drug Education Initiative" in March 2023 with $4.7 million to build guides for high-risk drugs like levothyroxine and warfarin. These aren’t one-size-fits-all anymore. They’re tailored. For thyroid patients: "Stick with one maker. Don’t switch unless your doctor says so." For blood thinners: "Your levels need checking every few weeks. That’s normal."
By January 2025, all Medicare Part D plans must give out patient materials that meet NIH health literacy standards. That means no more 12-point font, no more passive voice. Just clear, simple, direct language.
And it’s working. In 2023, CVS Caremark found that when patients got clear, medication-specific guides, switch-back rates dropped by 28%. That’s 28% fewer people going back to expensive brands because they were scared.

What should you do?
If you’re on a generic drug:
- Check the label. The active ingredient should match your old brand. If it doesn’t, ask.
- Don’t panic if the pill looks different. Color, shape, and size don’t change how it works.
- For thyroid, seizure, or blood-thinner meds: ask if your pharmacy uses the same generic maker each time. Consistency matters.
- Ask your pharmacist: "Is this an authorized generic?" If yes, it’s the brand made cheaper. No risk.
- Use the FDA’s "Generic Drugs" website. It’s updated quarterly. Free. Reliable. No ads.
If you’re switching from a brand to a generic and feel off? Don’t assume it’s the drug. Talk to your pharmacist. Maybe you switched from an authorized generic to a regular one. Maybe your body just needs a week to adjust. Or maybe-rarely-there’s a real issue. Either way, don’t quit the medicine. Ask first.
Where to find trusted guides
- FDAs "Generic Drugs" webpage - Updated quarterly. Includes charts like "What’s In a Name?" showing chemical names, generic names, and brand names side by side.
- Academy of Managed Care Pharmacy’s consumer toolkit - Free downloadable guides for common medications.
- Pharmacy chain handouts - CVS, Walgreens, and Walmart now use standardized templates based on FDA guidelines. Ask for one when you pick up your script.
- People’s Pharmacy podcast - Has 17 episodes on generic drugs. Real patient stories. No jargon.
Generic drugs saved the U.S. healthcare system nearly $2 trillion between 2009 and 2019. They’re not just cheaper. They’re just as safe. Just as effective. And yet, 38% of Medicare patients still think they’re weaker. That’s not about the medicine. It’s about the message. And that message is finally changing.


Holley T
February 24, 2026 AT 18:00Ashley Johnson
February 26, 2026 AT 05:01tia novialiswati
February 27, 2026 AT 20:30Christopher Brown
February 28, 2026 AT 19:16Sanjaykumar Rabari
March 1, 2026 AT 03:41Kenzie Goode
March 1, 2026 AT 05:59Dominic Punch
March 2, 2026 AT 10:54Valerie Letourneau
March 3, 2026 AT 06:53Khaya Street
March 4, 2026 AT 21:32Brooke Exley
March 5, 2026 AT 21:48